1.1 Background
In accordance with the World Health Organization, an adverse drug reaction is an unintentional and harmful reaction to a medicine that happens at doses typically utilized for disease prevention, identification, treatment, or modification of physiological function. One of the major contributors to illness, death, and rising healthcare expenses is adverse drug reactions (ADRs) [1]. ADRs significantly increase the cost of hospitalizations and other types of healthcare for the general public. ADRs are the fourth to sixth most prevalent cause of mortality in hospitalized patients, according to numerous research studies, as well as show consequences on illness, death, and economic trouble in society. [2].
According to the global scenario, ADRs account for 3% to 6% of hospital admissions, while the proportion of patients who suffer ADRs while hospitalized ranges from 1.5 to 35%. The average daily treatment fee per patient hospitalised with an Adverse Drug Reaction was projected to be Rs. 4945 [3]. However, the prevalence of ADRs in community settings is estimated to be 57%, and many of these cases go unreported by medical staff [4].
The foundation of several national Pharmacovigilance programs’ evaluation of drug safety data is the spontaneous reporting system (SRS) of ADRs. Spontaneous ADR reporting is a straightforward, inclusive, and cost-effective approach to pharmacovigilance, especially for novel medications, but its effectiveness is insufficient due to underreporting [5]. Many national Pharmacovigilance programs allow all healthcare workers, including pharmacists, to report potential ADRs [6]. Underreporting of ADRs has been a significant obstacle to enhancing Pharmacovigilance activities despite numerous attempts. One of the causes of underreporting may be healthcare practitioners’ poor comprehension of the significance of ADR reporting.
Various nations have their own reporting protocols to detect and report Adverse Drug Reactions, and doctors, apothecaries, and nurses all actively participate in this process. Pharmacists play a part in medication safety by helping to identify, prevent, report, and record adverse drug reactions (ADRs). Pharmacists have contributed to their respective national pharmacovigilance programs by reporting ADRs in nations including Spain and Portugal (26% and 24%), Canada (89%) and The Netherlands (40%), Australia (41%), The Netherlands (40%), and Spain (89%) [7]. Pharmacists are urged to report ADRs in the majority of developed nations. The excellence of Adverse Drug Reactions-reporting druggists is on the same level as other healthcare professions, according to a systematic examination of Adverse Drug Reaction-reporting methods in diverse nations. Pharmacists in India are not sufficiently aware of the Adverse Drug Reaction reporting mechanism, nevertheless. Clinical pharmacists are now in charge of tracking and reporting ADRs with the establishment of academic programs in pharmacy practice [8].
Most of the population considers community apothecaries to be the most approachable healthcare providers for all health service-associated problems [9]. In the past few decades, healthcare professionals have expanded their traditional tasks, such as producing and dispensing prescriptions, affecting the prescribing procedure, and providing pharmacological care amenities, and have become substantially more involved in patient care. Pharmaceutical care, according to Hepler and Strand, is the responsible administration of medication therapy with the aim of attaining certain results that enhance a patient’s standard of life. Due to their frequent contact with patients, community pharmacists can be valuable contributors to the pharmacovigilance program [10]. No medication is completely exempt from side effects, which is a well-known fact. The European Commission’s report on ADRs is shown in Table 1.1.
Table 1.1: European Commission report based on the ADRs
| patient’s statement | Report in percentage |
| Patients admitted to hospital | 0.3% – 5% |
| Report of the patient’s mortality | 3.5% |
| ADRs reported patients while they were in the hospital | 1.9% -6% |
Pharmacists employed in a community setting are in a distinctive position to be associated with monitoring and reporting ADRs related to non-prescription and complementary medicines. Community pharmacists’ contributions to the detection and reporting of ADRs to national Pharmacovigilance programs have been recognized in numerous international studies. The results of research performed in the Netherlands indicate that community apothecaries are responsible for 40% of the ADR reports that Lareb, the country’s official pharmacovigilance program, receives. It is crucial to keep community pharmacists’ wisdom, boldness, and motivation concerning Adverse Drug Reaction reporting up to date in order to increase the reporting rate [11].
This chapter discusses ADR monitoring within the community of pharmacies. It addresses the fundamental concepts and purpose underlying pharmacovigilance. The term ADRs and their enormous significance in the field of community pharmacy practice are thoroughly examined. This chapter places a strong emphasis on the vital importance of discovering and efficiently managing drug-induced side effects. It also addresses the thorough diagnosis procedure for ADRs, which includes both the tests and the important aspects that need to be considered. It also emphasizes the reporting of ADRs, which is an important step in the prevention of such adverse consequences. Additionally, examining the various barriers that community pharmacists may encounter in fulfilling this crucial role.
1.2 Pharmacovigilance (PV)
The pharmaceutical industry has a speciality called pharmacovigilance that focuses on monitoring and assessing the security of pharmaceuticals and other medical items. It entails the methodical gathering, examination, and reporting of negative events as well as other safety-related data connected to the use of pharmaceutical products. Since it was first introduced in the 1970s, “pharmacovigilance”—along with “drug safety”—has come to be used to refer to this area of study.
Pharmacovigilance is tasked with recognizing adverse events that go beyond the scope of a drug’s therapeutic impact. Identifying which side effects are acceptable to people in relation to how successfully they treat a disease. For example, eradicator is acknowledged to have some extremely dangerous adverse consequences, but when treating cancer patients who are in danger of losing their lives, these adverse effects are accepted due to the possibility of curing the patient [12]. However, if a medication prescribed to treat a headache resulted in identical adverse symptoms, the danger to the particular patient would be regarded as excessive, and the advantage would be considered insufficient to compensate for the potential harm.
With the introduction of standardized adverse event reporting, signal-detecting activities, and risk management strategies, pharmacovigilance has advanced over time. It is essential for maintaining the continued safety of pharmaceutical items and defending the general populace’s health.
A specific branch of science known as pharmacovigilance is described as follows:
“The science and practices involved in the identification, evaluation, comprehension, and mitigation of side effects or any other drug-related issue” [13].
1.2.1 Importance of Pharmacovigilance in ADR
The following list highlights the significance of pharmacovigilance in ADRs.
In order to detect and keep track of ADRs correlated with the usage of pharmaceutical products, pharmacovigilance is essential.
It helps to protect patient safety by identifying and assessing ADRs, especially significant and unexpected reactions.
Pharmacovigilance efforts enable the gathering and analysis of ADR data from medical professionals, patients, and pharmaceutical corporations. Examples include spontaneous reporting systems and databases.
Pharmacovigilance allows regulatory organizations to evaluate the safety profile of pharmaceuticals and implement the necessary regulatory actions, such as warnings, contraindications, or even the elimination of a drug from the marketplace, as needed.
Pharmacovigilance is indispensable for signal detection, which includes recognizing developing hazards or potential safety issues with certain medications.
Pharmacovigilance minimizes patient harm by tracking and analyzing ADRs, which helps to continuously enhance drug safety [14].
1.3 Adverse Drug Reaction
Adverse drug responses (ADRs), also known as ADEs or adverse drug effects, are unfavourable reactions or side effects that can happen when a pharmaceutical drug or medication is employed. ADRs can range in severity from moderate, manageable side effects to severe, perhaps fatal reactions. These reactions, which are separate from the medication’s intended therapeutic benefits, can happen when a drug is taken at the appropriate dosage [15].
The WHO created the conventional definition of an ADR in 1973, and it is as follows:
“Any adverse medication reaction that happens when a medicine is taken at dosages that are typically used in people to prevent, diagnose, treat, or modify illness or physiological function”.
An adverse drug response, occasionally known as an ADE, is a subtype of the more general opposing occurrence that is not just restricted to drug-related events but likewise overlays with certain kinds of drug mistakes. ADRs are unanticipated and adverse reactions to medications that happen at recommended therapeutic levels and are caused by the pharmacological characteristics of the medication itself. Usually, when a medicine is taken as directed, symptoms like nausea or skin rash occur. Contrarily, adverse drug events (ADEs) cover a wider range of negative effects associated with medicine use. While ADEs comprise occurrences resulting from drug errors, overdoses, or off-label usage, ADRs are a subset of ADEs [16].
For instance, if a patient is erroneously given the wrong drugs at the pharmacies and suffers adverse effects, that’s an ADE. Consider a patient going through intense itchiness and hives (ADR) after taking a prescribed antibiotic, which is ADR. Examples of ADR & ADE are presented in Figures 1.2 (a) as well as 1.2 (b).
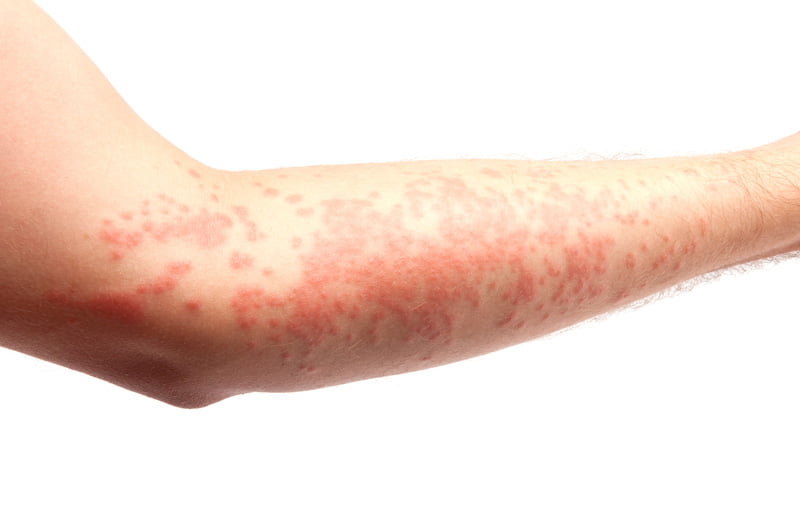
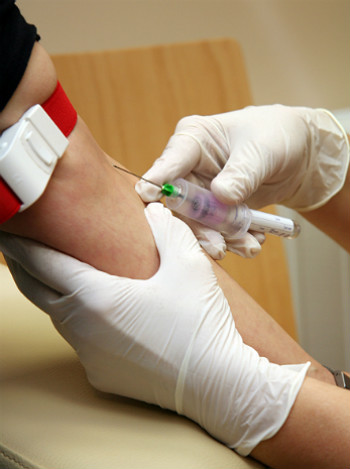
Figure 1.2 (a): Adverse drug reaction Figure 1.2 (b): Adverse drug events
1.3.1 Importance of ADRs
Based on the studies, ADRs represent between 2% and 6% of all hospital admissions. According to a recent meta-analysis, the 4th and 6th most common causes of mortality in the United States are ADRs. Recent surveys have also demonstrated the link between adverse medication events and longer hospital stays, which drives up the cost of treatment per patient [17].
Adverse medication reactions can have a variety of direct and indirect effects, including:
Negative outcomes for patients’ quality of life
Attendance at a primary health centre or hospital admission
Extensive inpatient hospital stays
A rise in the price of patient care
The patient’s loss of trust in the treating physician
Negative responses can result in death, permanent disability, congenital anomalies, or birth abnormalities.
1.4 Classification of Adverse Drug Reaction
ADRs can be divided into a number of categories depending on a variety of factors, enabling a thorough comprehension of their nature and traits. Type A and Type B reactions are distinguished by one common classification. Type A reactions frequently emerge from a drug’s recognized pharmacological effects and are connected to its therapeutic effect. They are predictable and dose-dependent. This includes, for instance, gastrointestinal distress brought on by non-steroidal anti-inflammatory medicines (NSAIDs). Type B responses, on the other side, are unanticipated, peculiar, or hypersensitive reactions that are unrelated to a drug’s established effects. These can range from mild allergies to severe immune-mediated reactions.
Adverse Drug Reactions can also be categorized based on how bad they are (mild, modest, extreme), onset and duration (immediate, acute, subacute, or chronic), dose-relatedness (dependent or independent of dose), affected organ or system (cutaneous, hepatic, renal, haematological, cardiovascular, etc.), mechanism (pharmacological, immunological, idiosyncratic, metabolic), and time course (immediate onset or delayed onset). Effective identification, management, and reporting of ADRs by healthcare professionals is aided by this complex classification system, which ultimately improves patient safety and directs the regulatory and drug development processes [18]. Classification of adverse medication reactions is provided in Table 1.2. The different kinds of adverse medication reactions are depicted in Figure 1.2.
Table 1.2: Classification of ADRs
| Reaction Type | Mnemonic | Examples of Attributes | Examples |
| Dosage-related | Augmented |
|
|
| Non-Dosage-associated | Bizarre |
|
|
| Dose and time-correlated | Chronic |
|
|
| Time-related | Delayed |
|
|
| withdrawal | End of use |
|
|
| Unanticipated therapeutic failure | Failure |
|
|
1.5 Factors Mediating to Develop Adverse Drug Reactions
Medication-related risk factors, patient-related risk factors, and social factor categories are used to classify risk factors for adverse drug responses.
1.5.1. Medication-related risk factors
This section discovers the Medication-related risk elements that result in Adverse Drug Reactions, such as the Dose and frequency of the drug and polypharmacy.
1.5.1.1 Dose and frequency of the drug
The likelihood of adverse drug reactions can be greatly influenced by the drug dosage, frequency of organisation, as well as the moment of the day at which the medication is given. ADRs can arise when a medication is administered at subtherapeutic or hazardous doses, more frequently, or at different times of the day than is recommended. To avoid disrupting sleep, bendrofluazide’s diuretic action shouldn’t be taken at nighttime, although the antiplatelet action of the aspirin pill is better when taken at night [19].
1.5.1.2. Polypharmacy
It is recognized that individuals who take many medications are at risk for adverse drug responses. The risk of ADRs in elderly individuals rises steadily as the amount of medications utilized increases. Age and polypharmacy are the two main risk factors, according to an Indian study on adverse medication responses. Utilizing the least amount of medications necessary to treat a disease condition can lower the occurrence of ADRs. The enhanced ADR risk is multiplied by the medicine by 1.1 times for each extra dose. The combined toxic effects of two or more medications can be significantly more harmful than the effects of the individual medications [20]. When compared to other medications, the use of NSAIDs raises the incidence of peptic ulcers in elderly people by 10%. The risk of developing a peptic ulcer is increased by 15 times compared to persons who do not take any medications when NSAIDs and corticosteroids are used together. Anticoagulants raise the risk of hemorrhagic peptic ulcer by severalfold when combined with NSAIDs, while the risk is decreased when the medications are used alone [21].
1.5.2. Patient-related factors
This section discovers the Patient-related factors that lead to Adverse Drug Reactions (ADRs), including Age, Gender, Pregnancy, Fetal development, Renal function and Allergy.
1.5.2.1. Age
Patients over the age of 60 are more likely to experience negative pharmacological responses from their prescriptions. Compared to the other age groups, elderly persons had ADRs more frequently. According to a study, elderly adults are more prone to ADRs than younger persons. According to a New Delhi study on the monitoring of the adverse effects of antihypertensives, patients older than 40 were more likely to experience ADRs, and middle-aged patients and women made up the bulk of those who reported them. The increased frequency of ADRs could be attributed to the health issues that begin to manifest beyond the age of 40. Multiple drugs, both pharmaceutical and without a prescription, must be taken concurrently for certain health issues [22]. With age, the kidneys and the liver’s capacity to digest medications and eliminate them also declines. Negative effects are brought on by the medications’ increased body concentration due to the kidneys’ decreased ability to remove the substances from the body. Compared to young individuals, older adults have a higher percentage of body fat and less body water. Medicines that dissolve in fat accumulate in tissues and can lead to enhanced drug activity with accompanying negative effects when there is a low body water content and a higher body fat content [23].
1.5.2.2. Gender
The gender differences in adverse medication reactions are also present. Pharmaceutical ADRs are likely to be particularly prevalent in female patients. The anatomical and physiological makeup of males and females differs significantly. Compared to men, women often have larger auxiliary organs, higher fat deposits, and lesser body mass index. As an illustration, the hepatic enzyme CYP3A4, which metabolizes a variety of medicines, is more hyperactive in girls than in males when it comes to drug metabolism rates [24].
The biodistribution and pharmacological response of the drugs are impacted by the differences in anatomy and physiology between men and women. According to a study from north India on the relationship between ACE inhibitors and wheezing, women are more likely than manpower to experience coughing. Males and females differed markedly from one another in the occurrence of adverse drug responses related to cardiovascular drugs (54% vs. 46%), according to a study. Females were 4.02 times more likely than males to have an ADR occur. For cardiac glycosides and high ceiling diuretics, females 13 had a 2.38 times higher likelihood than males of developing ADR. However, a study revealed that Compared to men, women were far more inclined to report experiencing physical disease symptoms [25].
1.5.2.3. Pregnancy
Pregnancy has been proven to cause a number of biological transformations that alter the pharmaceutical and pharmacological effects of the drug, including changes in cardiovascular function. Increased heart rate (10–15 beats per minute), stroke volume, amounts of blood (1500–1800 ml), nephritic drug absorption due to augmented renal bloodstream (30%), increase in Glomerular Filtration Rate (50%) and reduction in serum protein (1–1.5) all contribute to an increase in cardiac output (32%) in this condition. Negative medication reactions have an effect on the pregnant woman, the fetus, or both during pregnancy. [26].
1.5.2.4. Fetal development
The fetus in the uterus is more sensitive to the medications in parental circulation as it is delicate in nature, and a lower level of metabolism and excretion can end up in teratogenicity. Teratogenicity and other unfavourable drug reactions during organ formation are caused by the medications used during the first trimester. The repeated administration of medications during the second or third trimester of pregnancy has been linked to haemorrhage, respiratory distress, illness, and development retardation. The harmful effects are dependent on the drug kind dosage administered, the length of time, and the developmental stage of the fetus [27].
1.5.2.5. Renal function
Kidney disorders affect drug clearance and metabolism, which causes toxicity and subtherapeutic effects of the medication. Creatinine clearance levels can be used to measure it. The drug transport system and metabolic enzymes alter in specific ways in patients with renal failure because of the lower drug clearance. ADRs from renal insufficiency are more likely to occur when there is further illness or infection.
1.5.2.6. Allergy
Cross-sensitivity develops between drugs in the exact same pharmacological category and with comparable chemical compositions, and medication-independent cross-reactive antigens have the potential to trigger sensitizations that result in allergies to drugs. The second drug exposure after the initial medication exposure leads the T-cells and antibodies to be stimulated and enter the elicitation phase, similar to type I to IV immunological reactions. Drug allergies that develop in patients are likely to be type I or type II reactions, but type II and III reactions are frequently unusual. Serum sickness, systemic lupus erythematosus, and vasculitis are a few examples of type III reactions. Sulphonamides and beta-lactam antibiotics are two more prevalent medications that cause drug allergies.
1.5.3. Social factors
This section explores the social factors that lead to ADRs, embracing race and ethnicity, in addition to alcohol and smoking habits.
1.5.3.1. Race and Ethnicity
The ethnic background is determined by genetics. It makes sense, given genetic variability, individual variations in the capacity of certain enzymes to metabolize medicines, and variations in drug receptors and transporters. The frequency of ADRs varies depending on the genetic differences across the races. Approximately 19% of the participants in a large population-based cohort trial with a sample size of 2225 subjects who got ACE inhibitors and were assessed for risk factors for adverse drug reactions were excluded due to ADRs. It was discovered that compared to other ethnic groups, African Americans were more prone to angioedema caused by ACE inhibitors [28]. According to the research that was done, black people had a 3.0 relative risk of angioedema and a greater risk of cerebral haemorrhage than the populations of other races. In comparison to white patients, East Asian patients are three times more likely to experience ADRs, particularly cough. Cases with Parkinson’s disease who take catechol-O-methyltransferase inhibitors are more inclined to encounter adverse drug reactions if they have the UDP-glucoronosyl transferase-1 A9 genotype. Patients who had greater levels of CYP1A2, fewer allele mixtures (8/12; 67%), and lesser levels of CYP1A2 – mRNA had higher ADRs than those who did not.
1.5.3.2. Alcohol
ADRs can develop in alcoholics because alcohol alters the metabolism of drugs and encourages their growth. Cirrhosis of the liver has an impact on medication metabolism, and research has shown that hepatitis C is strongly linked to both a decreased rate of drug metabolism and an increased risk of drug toxicity. When taking alcohol and NSAIDs at the same time, people with peptic ulcer disease or gastritis may experience internal bleeding. Patients who had taken medications and alcohol at the same time had ADRs such as nausea, vomiting, headaches, sleepiness, fainting, and hypotension [29].
1.5.3.3. Smoking
It is an independent predictor of cardiovascular illnesses, peptic ulcers, and cancer. Smoking causes the liver cytochrome P450 isoenzymes CYP1A1, 1A2, and perhaps 2E2 to be produced, which increases medication metabolism and reduces the pharmacological activity of the pharmaceuticals. According to the research, smokers’ blood pressure is affected less by beta-blockers. Nicotine interacts with several medications, including theophylline, insulin, H2 receptor blockers, oral contraceptives, and other medicines like thiothixene. Human studies have shown that smokers with insulin-dependent diabetes need 15–20% more insulin than non-smokers, and heavy smokers may need up to 30% more [30].
1.6 Common Adverse Drug Reactions (ADRs)
Common ADRs are unwanted consequences or adverse reactions that can happen when people take specific medications as directed. These ADRs frequently appear in the package insert or drug labelling of a prescription and are typically expected and known by healthcare professionals. Even while common ADRs are frequently less severe than uncommon or major adverse effects, they can nonetheless have an influence on patients’ well-being and demand care. Examples of common ADRs are,
Angioedema of Lips
Erythema Multiforme
Stevens-Johnson Syndrome (SJS)
Ampicillin-Induced Drug Eruption
1.6.1 Angioedema of Lips
Angioedema is a disorder that causes rapid swelling in the deeper skin layers, frequently affecting the lips, eyes, and face. Certain drugs, especially ACE inhibitors like lisinopril that block the angiotensin-converting enzyme (ACE), can contribute to it. Although ACE inhibitors are frequently used to treat hypertension and heart failure, some people may experience angioedema as an adverse drug reaction (ADR) [31].
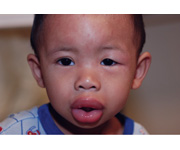
Figure 1.4 Angioedema of Lips
1.6.2 Erythema Multiforme
A skin condition known as erythema multiforme is distinguished by unique target-like skin lesions with concentric rings. It is an ADR and can be brought on by drugs, infections, or other things. Skin rashes are a common symptom of erythema multiforme, which can also affect mucous membranes and cause painful ulcers.
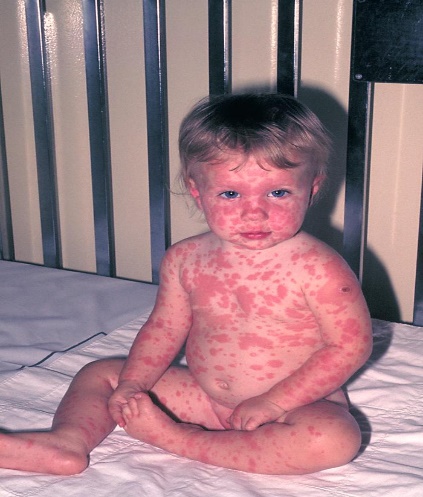
Figure 1.5 Erythema Multiforme
1.6.3 Stevens-Johnson Syndrome (SJS) by Phenytoin
A serious and perhaps fatal skin disorder is Stevens-Johnson Syndrome. It occurs as an ADR with a number of medications, including antiepileptic treatments like phenytoin. Flu-like symptoms are frequently the first sign of SJS, which then lead to the emergence of painful blisters and extensive skin separation, signifying an ADR. It is regarded as a medical emergency and needs to be treated right away.
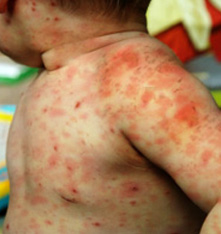
Figure 1.6 Stevens-Johnson Syndrome
1.6.4 Ampicillin-Induced Drug Eruption
An antibiotic called ampicillin is used to treat several bacterial infections. Drug eruptions, which are skin rashes or lesions brought on by a hypersensitivity reaction to the treatment, can nonetheless occasionally develop from it. These eruptions, which are considered ADRs, can range in intensity and involve symptoms including itching, redness, or blistering.
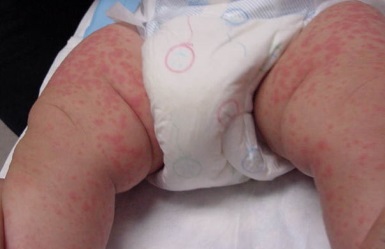
Figure 1.7 Ampicillin-Induced Drug Eruption
1.7 Proclamations of Unwanted Drug Effects
According to United States Food and Drug Administration data, the last decade saw an almost two-fold increase in drug side effects. It has been noted that a sizeable number of patients were discharged as a result of the severe side effects of medications. In the 10th problem of the Archives of Internal Medicine, this account has been verified. Between 1998 and 2005, the number of reports per year improved up to 2.6-fold. The number of reported adverse events grew from 34,966 in 1998 to 89,842 by 2005 [32].
Tables 1.3, 1.4, and 1.5 show, respectively, the record of the number of pharmaceuticals on the market that cause ADRs, a record of drugs that are prohibited, and a list of drugs that were withdrawn after ADRs were seen in the patient.
Table 1.3: Records of the number of Drugs in the market that produce Adverse Drug Reactions
| Introduced drug | Different kinds of dosage form | Afflicted patients | Negative effects |
| Oxycodone | Tablet-film coated and extended-release | 5% | Constipation, nausea, somnolence, dizziness, vomiting, sweating, asthenia, headache |
| Fentanyl | IV, patch | 7.9% | Skin reactions, respiratory depression, mental changes, stroke |
| Clozapine | Tablets | 2.7% | fever, body aches, cough, sore throat, rapid heart rate, |
| Methadone | Tablets | 3.6% | Respiratory depression, cramps, nausea, dilated pupils, |
| Morphine | Injection | 3% | Constipation, addiction, depression, renal failure, slow heartbeat, |
Table 1.4: Records of banned drugs
| Promoted drug | Dosage form | Banned date | Causes | Death occurred |
| Mibefradil (Posicor) | Tablets | June 1998 | hypertension, chronic angina, rhinitis, heart stroke, | 123 deaths in 1 year |
| Astemizole (Hismanal) | Tablets, capsules | July 1999 | Heart problems, death, cardiac arrest, cardiac arrhythmias, bradycardia, | 1-2% |
| Cisapride (Populsid) | Tablets, oral suspension, capsules, medi-melt tablets, injections | January 2000 | Fast heartbeat, convulsions, irregular heartbeat, QT prolongations, | 80 deaths during a clinical trial |
| Rofecoxib | Tablets | November 2007 | abdominal pain, tenderness, or discomfort, nausea, blood while vomiting, unexplained weight gain, | 2-3% |
Table 1.5: Records of drugs that were eliminated after ADRs were discovered in patients
| Marketed drug | Dosage form | Eliminated due to | Affected patients |
| Mibefradil (Posicor) | Tablets | Reported to cause drug interaction | 123 deaths in 1 year |
| Cisapride | Tablets, oral suspension, capsules, medi-melt tablets, injections | Caused cardiac arrhythmias | 80 deaths during a clinical trial |
| Thalidomide | Capsules | Risk of teratogenicity | 6-8% |
| Nimesulide (below 13 years of age) | Tablets, oral suspension, gel, suppositories | Caused life-threatening hepatotoxic effects | 1.3% |
| Gatifloxacin | Tablets, injectables | Causes hyperglycemia and liver damage | 1.2% |
1.8 Diagnosing Adverse Drug Reaction
In the identification of negative drug reactions in a hospital context, patients can be contacted based on newly appearing or increasing symptoms, which may be the first indication of ADR. Patients frequently ask the pharmacist in a community pharmacy for assistance on how to treat different ailments in the household. This is a chance for the druggist to ascertain the cause of ADRs. Drugs like antibiotics are linked to ADRs. Patients frequently question the nurse or doctor about a new symptom that necessitates the pharmacist in an inpatient setting. A thorough investigation of the new symptom and how it is being treated may reveal medication therapy or further ADR symptoms. A laboratory test can be performed on the medications that cause ADR.
Serum drug levels alert the doctor to ADRs brought on by drug toxicity or unsuccessful treatment attempts. A laboratory test can both establish the normal organ function and rule out an alternate diagnosis. When using a novel medicine, the ADR might be determined by the initial laboratory tests. ADR may not always be indicated by abnormal laboratory results, but they do enable the practitioner to closely examine the patient and make that determination [33]. A less visible strategy for the diagnosis of ADR is the examination of the drug order in both hospitalization and outpatient treatment. The abrupt, unexpected withdrawal of a medication or a significant dosage adjustment can be used to detect ADRs. Naloxone, Flumazenil, Diphenhydramine, antiemetics, vitamin K, sodium polystyrene sulfonate, corticosteroids, or diarrhoeas are a few examples of medications that can be used to treat ADRs.
The comprehensive procedure outlined below may be beneficial when assessing possible adverse medication reactions:
Step 1: Finding out which drugs the patient is currently taking
Step 2: Confirm whether the symptoms and indications persisted following the start of the medicinal intervention.
Step 3: Finding out how much time has gone between the beginning of the drug treatment and the development of the symptoms
Step 4: Stop using drugs and monitor symptoms and warning signs.
Step 5: In rare cases, it could be required to restart medication therapy while keeping an eye out for the reappearance of signs and symptoms.
1.9 Reporting adverse drug reactions
Community pharmacists’ reporting of ADRs may provide useful data about the drug’s safety profile. Using this information will make it easier to develop measures that will reduce the likelihood of ADRs occurring in community settings.
1.9.1 Indian Scenario
Community pharmacy is not regarded as a well-established medical speciality in India. Most of the neighbourhood pharmacies in this country are run by pharmacists with diplomas who are underqualified (in comparison to many other nations). The role of pharmacists in promoting health and ensuring the safe and effective practice of medications has been envisioned by the WHO for some time. It is uncertain at this time whether Indian community pharmacists participate in these initiatives. In India, community pharmacy practice is not given enough attention. The activities of community pharmacists need to be studied, and their education and training need to be routinely evaluated. Because of a lack of awareness, poor reporting by general practitioners, and a lack of a reporting mechanism, the majority of prescriptions are filled in the community setting [34].
Community pharmacists in India are only allowed to practice retail pharmacy; they are not allowed to manage or provide consultation regarding medication therapy. As the patients’ last source of contact, they can be crucial in the identification and reporting of possible ADRs. In India, it is typical for the vast majority of medications to be given out without a prescription. Medications that frequently have side effects (such as hypnotics and non-steroidal anti-inflammatory meds) are given out without a doctor’s advice. Therefore, there should be a need for regulatory and instructional measures to enhance community pharmacists’ knowledge and professional conduct. Clinical pharmacists in India have assumed a leadership role in supervising and reporting ADRs, structured patient education, and patient counselling, among other things, as a result of the changing landscape of pharmacy practice and the advent of clinical pharmacy programs.
Despite being able to report, community pharmacists’ reporting rates have remained low as compared to those of other healthcare providers. To strengthen the ADR reporting system in India, several research should be done to evaluate the attitudes and perceptions of community pharmacists concerning ADR. Therefore, community pharmacists can be crucial in the identification and reporting of any Adverse Drug Reactions. Communal apothecaries have been able to report, but their reporting rates have remained low when compared to those of other healthcare providers [35].
1.9.2 Barriers to Reporting the ADRs by the Community Pharmacists
Community pharmacists’ understanding of ADR monitoring and reporting activities is still developing in India. The main cause of this is that community pharmacies are frequently run by people who lack training and qualifications. Additionally, registered pharmacists working in and managing pharmacies lack sufficient knowledge about adverse drug reactions and how to report ADRs. Also absent from the curriculum is any mention of pharmacovigilance efforts. In India, pharmacists’ services are also not permitted, and they are not in charge of overseeing drug therapy. Pharmacists are, therefore, not particularly interested in Pharmacovigilance efforts. Patients’ final point of contact is the neighbourhood drugstore, where they buy their medications and discuss any issues they have with their drug regimen. Most of the time, pharmacists are not physically present to answer patients’ questions [36].
As a result, people don’t seem interested in telling the pharmacist about any negative events. Healthcare providers’ ignorance of the significance of ADR reporting, the low percentage of staff educated in pharmacovigilance, and other factors have also been noted as barriers to ADR reporting. Lack of priority setting within the drug regulating authority and inadequate emphasis on pharmacovigilance in public health initiatives Lack of facility-based technical and financial resources for data collection and analysis Inconsistent distribution and collection of ADRs forms from healthcare facilities are caused by a lack of organizational strength at the agency responsible for regulating medications. Absence of consistent monitoring and oversight by the pharmacovigilance coordinator at the drug regulatory organization [37].
According to a study conducted by Toklu HZ in Istanbul, failure to report ADRs was brought on by an absence of time, a disparity in care priorities, unpredictability regarding the drug that led to the ADR, difficulty obtaining forms, ignorance of the reporting criteria, and an absence of knowledge of spontaneous reporting systems. Based on a survey given to the staff, the criteria that prevent ADR reports are depicted in Table 1.6, and the graphical representation of Factors that hinder ADR Reporting is shown in Figure 1.7 [38].
Table 1.6: Factors that impede Adverse Drug Reaction Reporting
| Factors that make you reluctant to report Adverse Drug Reactions | Total no. of contributors n=151 (%) |
| not sure how to report | 14(9.2%) |
| Don’t consider it important. | 3(1.9%) |
| Taking care of patients came first | 30(19.8%) |
| Access to the ADR reporting form is not available | 23(15.2%) |
| problem with patient confidentiality | 54(35.7%) |
| The duty of physicians is to report ADRs | 0(%) |
| It takes time to report | 13(8.6%) |
| Problem with liability | 2(1.3%) |
| All of above | 12(7.9%) |
1.10 Monitoring of ADRs
The practice of routinely monitoring any unwanted effects brought on by taking any medicine is known as ADR monitoring. Tracking ADRs requires an essential component called pharmacovigilance. The law mandates that pharmaceutical regulators track any potential undesirable responses as well as the market for their products. ADRs can be caused by the use of several pharmaceuticals, including herbal medicines, skincare products, medical supplies, and biology. This monitoring procedure is used to guarantee that patients receive efficient and secure drugs [39].
If any adverse occurrences are not disclosed, medicinal products may have harmful and dangerous effects. Thus, properly establishing ADR monitoring programs will help to reduce the side effects of pharmaceuticals.
1.10.1 Need for the ADR monitoring
A pharmaceutical medicine normally goes through extensive clinical trials to assess its safety and efficacy prior to being released on the market. These trials are meticulously planned studies with a chosen few patients that are carried out under carefully monitored conditions. The main goal of these trials is to obtain information about the drug’s performance in a controlled setting and whether it provides the desired therapeutic advantages [40].
Clinical trials in a controlled environment can have certain drawbacks, though. The participants taking part in these trials frequently represent a certain subgroup of the population that has been carefully selected to meet particular requirements. The complete range of variation that exists in the actual world is not captured by this controlled environment, but it does allow researchers to isolate and investigate the effects of the medicine under perfect circumstances.
A medicine may experience new and unanticipated side effects in addition to its intended therapeutic efficacy after it is put on the market and given to a larger and more varied spectrum of patients. Adverse Drug Reactions (ADRs) are the name given to these side effects. ADRs can include minor, controllable side effects as well as serious, perhaps fatal, consequences.
1.10.2 Benefits of ADR monitoring
The following advantages can be obtained through an ADR monitoring and reporting program:
It provides details about the reliability and security of medicinal items.
Plans for risk management are started.
It assists in assessing the occurrence of ADRs and prevents the predictable harmful effects.
It raises awareness of ADRs and educates the medical staff, patients, pharmacists, and nurses about adverse drug reactions.
ADR monitoring’s primary goals are to disclose the type, frequency, and risk factors that can result in adverse reactions.
1.11 Proactive Strategies for Preventing ADRs in Community Pharmacies
It is vital to prevent ADRs in the communal pharmacy embarking to guarantee patient security as well as successful medication results. While some ADRs, such as anaphylactic reactions following prior unremarkable exposures, can be unanticipated, many are really avoidable with a well-thought-out strategy. In this context, the idea of preventability is directly related to the medication treatment plan’s alignment with current evidence-based guidelines and customization to the specific patient’s needs [41].
Two fundamental steps can be followed to proactively prevent ADRs:
Recognizing Susceptibility
The risk of ADRs can be reduced, and prescribing decisions can be made with greater precision by having a complete awareness of patient susceptibilities. To prevent re-exposure to the offending medicine, a patient’s prescription history should be carefully examined to find any prior ADRs. ADR risk can also be predicted by taking into account a number of susceptibility characteristics, including age, gender, pregnancy status, and ethnicity. For instance, different ethnic groups may be more or less susceptible to particular ADRs, necessitating the use of customized medications. By identifying which patients are more likely to experience a certain ADR, emerging fields like pharmacogenetics offer prospects for more individualized therapy.
Treatment Strategy Improvement
ADRs can be decreased in part by using cautious and safe prescribing procedures. Plans for treatment must not only take into account and minimize any negative effects but also include targeted therapeutic results. For instance, supplementing methotrexate with folic acid lowers the possibility of side effects associated with a folate deficiency, and cautious tracking of electrolytes and renal function is required when using particularly active medicines or diuretics. It’s crucial to remember that cautious prescribing occasionally entails choosing non-pharmacological or conservative therapy modalities.
A systemic approach is required to implement a complete plan in community pharmacy to prevent ADRs. This method uses a variety of tactics, such as strong communication between medical staff members and actively involving patients in their care. Pharmacists can get help from clinical decision support systems at the time of care by getting patient-specific warnings and monitoring suggestions to lower the risk of harm. As various systems differ in how they deliver information, it’s crucial to establish a balance between using decision help and avoiding alert fatigue. Community pharmacists can greatly reduce the occurrence of preventable ADRs and promote safer drug practices by working with healthcare professionals and involving patients in their care.
1.12 Pharmacists
Pharmacists are people with specific education and training in pharmacy, which includes having a complete understanding of pharmaceuticals, their effects on the body, and the guidelines for using medications safely and effectively. They are qualified healthcare professionals who are in charge of making sure that patients get the correct drugs in the right amounts and with the right usage instructions.
1.12.1 Role of Pharmacists in ADR Monitoring
In the realm of healthcare, druggists hold a vital part as vigilant guardians of patient safety. They have the knowledge and skills necessary to properly monitor and manage Adverse Drug Reactions (ADRs) thanks to their training and competence in pharmacy. Pharmacy professionals are at the forefront of ensuring that patients receive the proper medications, are provided with the proper dosages, and have clear instructions. They provide patients with counselling that goes beyond dispensing, enlightening them about potential ADRs and the value of swiftly reporting them. Pharmacy professionals perform rigorous drug reviews, looking for possible interactions and contraindications that could result in ADRs. They also monitor patients on long-term drugs, evaluate patient reactions, and identify both typical and unusual adverse drug reactions (ADRs) [42].
They work closely with medical professionals, contribute to ADR reporting systems, and actively participate in patient education, promoting open dialogue about issues relating to medications. In order to improve overall healthcare quality and patient safety, pharmacists play a crucial role in ADR monitoring through their constant dedication to lifelong learning. The following is a list of pharmacists’ responsibilities,
Medication Dispensing
Patients receive prescription and non-prescription drugs from pharmacists. They make sure the medication is what was prescribed, provide patients explicit instructions on how to take the medication, and even provide patients with counselling regarding potential ADRs.
Medication Review
In order to discover possible drug interactions, allergies, or contraindications that could result in ADRs, pharmacists analyze patients’ prescription profiles. When necessary, they may work with healthcare professionals to modify medication schedules.
Monitoring and Assessment
Pharmacists watch patients on long-term medicines. They monitor changes in vital signs and laboratory findings, evaluate how well patients respond to treatment and look for any clinical symptoms that might point to ADRs. The success of early detection and intervention depends on this attention.
ADR Recognition
Pharmacists are taught to spot conventional ADRs and unusual symptoms that could point to a drug-related problem. Their knowledge identifies possible issues and directs proper action.
Reporting
It is the duty of pharmacists to notify the appropriate regulatory bodies or pharmacovigilance programs of suspected ADRs. These studies advance our knowledge of pharmaceutical safety and may prompt regulatory action.
Collaboration
To discuss medication-related issues, improve treatment regimens, and ensure coordinated patient care, Other health care providers, including nurses and doctors, collaborate with dispensers.
Documentation
Pharmacists keep thorough records of each patient’s prescription history, including ADRs and the results of those events. Future decision-making is aided by this knowledge, which also improves patient security.
1.13 Problem statement
Existing problems in Adverse Drug Reaction (ADR) monitoring typically include underreporting by healthcare professionals, deficiency of awareness and absence of knowledge about ADR reporting, limited communication between healthcare settings, and inadequate resources for systematic ADR data collection and analysis [34]. Similar issues, like low pharmacist awareness and education, a lack of standardized reporting methods, a lack of time and resources, and worries about potential liability, occur in community pharmacies [35][36]. To address these issues, ADR Monitoring in Selected Community Pharmacies is proposed. This significantly reduces underreporting by utilizing their accessibility to healthcare seekers, knowledge of drugs, and proximity to patients. It improves community pharmacists’ knowledge and reporting abilities through educational interventions and specialized training programs, which leads to an increase in ADR awareness and reporting practices. Additionally, by implementing a standardized reporting system inside community pharmacists, this project promotes improved data exchange and communication between healthcare settings, enabling thorough ADR monitoring. Overall, this strategy improves ADR monitoring by utilizing community pharmacists’ latent capacity and strengthens the pharmacovigilance system.
1.14 Motivation of the study
The motivation to perform this study arises from the urgent requirement to address the persistent obstacles and inadequate reporting of ADRs within the community pharmacy background. ADRs can present significant threats to patient safety, and the existing deficiencies in ADR monitoring, such as low pharmacist awareness, a lack of standardized methods, and resource limitations, require an alternative approach. This study intends to make use of community pharmacists’ distinctive position as healthcare professionals to increase ADR awareness and reporting habits and ultimately improve patient safety by involving them in ADR monitoring through education, training, and structured reporting systems. This study seeks to cover the ADR surveillance discrepancy in neighbourhood pharmacies, strengthening the pharmacovigilance system and improving patient outcomes overall.
1.15 Objective of the study
General Objective
To design, implement and evaluate the ADR reporting system by selected community Pharmacists.
Specific Objectives
To evaluate how the educational interventional programs have affected the practices, attitudes, and understanding of community pharmacists regarding Adverse Drug Reaction reporting.
To establish the training manual and evaluate how well it helped community pharmacists recognize and report ADRs.
To determine the obstacles experienced by trained community pharmacists when reporting and monitoring ADRs.
To develop and implement techniques to enhance ADR reporting by trained community pharmacists.
1.16 Thesis Organisation
The first part of this chapter is the background: Pharmacovigilance (PV), Adverse Drug Reaction, Classification of ADRs, Factors Mediating the Development of Adverse Drug Reactions, Common Adverse Drug Reactions (ADRs), Proclamations of adverse effects of drugs, Diagnosing ADR, Monitoring of Adverse Drug Reactions, Proactive Strategies for Preventing ADRs in Community Pharmacies, Pharmacists is addressed in this thesis. The second part of this chapter provides an overview of a Problem statement, the Motivation of the study, the Objective of the study, and the Summary. Chapter 2 conducts a comprehensive literature review on ADR monitoring. It focuses on the responsibilities of neighbourhood pharmacists while exploring the historical and worldwide context of ADR monitoring. The chapter discusses earlier studies and research while emphasizing the value of standardized reporting practices.
1.17 Summary
Adverse Drug Reactions (ADRs) Monitoring within the Community of Pharmacies has been addressed in this chapter. It examined the fundamental ideas and motivation behind pharmacovigilance. The analysis went in-depth on the idea of ADRs and their enormous importance in terms of community pharmacy practice. This chapter placed a strong emphasis on the critical necessity of identifying and effectively managing drug-induced side effects. Additionally, it included the thorough diagnosis procedure for ADRs, which included both diagnostic instruments and the important aspects that needed to be considered. It also covered the reporting of ADRs, which was an important part of preventing such adverse occurrences. It also covered the numerous obstacles that community pharmacists could run into when assuming this crucial job.
Reference
Zhao, Y., Wang, T., Li, G. and Sun, S., 2018. Pharmacovigilance in China: development and challenges. International journal of clinical pharmacy, 40, pp.823-831.
Thomas, T.M., Udaykumar, P. and Scandashree, K., 2013. Knowledge, attitude and practice of adverse drug reaction reporting among doctors in a tertiary health care centre in South India. International Journal of Pharmacology and Clinical Sciences, 2(3). 5
Rajakannan, T., Mallayasamy, S., Guddattu, V., Kamath, A., Vilakkthala, R., Rao, P.G. and Bairy, L.K., 2012. Cost of adverse drug reactions in a South Indian tertiary care teaching hospital. The Journal of Clinical Pharmacology, 52(4), pp.559-565. 6
D’Cruz, S., Sachdev, A. and Tiwari, P., 2012. Adverse drug reactions & their risk factors among Indian ambulatory elderly patients. The Indian Journal of Medical Research, 136(3), p.404. 7
Inácio P., Cavaco A., and Airaksinen M. (2017) The value of patient reporting to the pharmacovigilance system: a systematic review. Br J Clin Pharmacol, 83: 227–246 8
Kalaiselvan V, Prasad T, Bisht A, Singh S, Singh GN. Adverse drug reactions reporting culture in pharmacovigilance programme of India. Indian J Med Res 2014;140:563-4 9
Pant, J., Marwah, H., Singh, R.M. and Hazra, S., 2021. An overview of the worldwide master key for pharmacovigilance and its role in India. Journal of Pharmacovigilance and Drug Research, 2(2), pp.19-26.
Ahmad, A., Patel, I., Balkrishnan, R., Mohanta, G.P. and Manna, P.K., 2013. An evaluation of knowledge, attitude and practice of Indian pharmacists towards adverse drug reaction reporting: A pilot study. Perspectives in clinical research, 4(4), p.204. 13
Ma’aji, H.U. and Ilyas, O.S., 2014. Assessment of knowledge, attitude and practice of community pharmacists towards pharmaceutical care in Kaduna State, Nigeria. Int J Pharm Teach Pract, 5(2), pp.972-976. 14
Olowofela, A., Fourrier-Réglat, A. and Isah, A.O., 2016. Pharmacovigilance in Nigeria: an overview. Pharmaceutical Medicine, 30, pp.87-94.
Hanafi, S., Torkamandi, H., Hayatshahi, A., Gholami, K. and Javadi, M., 2012. Knowledge, attitudes and practice of nurse regarding adverse drug reaction reporting. Iranian Journal of Nursing and Midwifery Research, 17(1), p.21.
Beninger, P., 2018. Pharmacovigilance: an overview. Clinical therapeutics, 40(12), pp.1991-2004.
Jha, N., Shankar, P.R., Bajracharya, O., Gurung, S.B. and Singh, K.K., 2012. Adverse drug reaction reporting in a pharmacovigilance centre of Nepal. The Australasian Medical Journal, 5(5), p.268.
Olsson, S., Pal, S.N. and Dodoo, A., 2015. Pharmacovigilance in resource-limited countries. Expert review of clinical pharmacology, 8(4), pp.449-460.
Giardina, C., Cutroneo, P.M., Mocciaro, E., Russo, G.T., Mandraffino, G., Basile, G., Rapisarda, F., Ferrara, R., Spina, E. and Arcoraci, V., 2018. Adverse drug reactions in hospitalized patients: results of the FORWARD (Facilitation of Reporting in Hospital Ward) study. Frontiers in pharmacology, 9, p.350.
Chen, L., Jiang, L., Shen, A. and Wei, W., 2016. Development of a quality instrument for assessing the spontaneous reports of ADR/ADE using Delphi method in China. European Journal of Clinical Pharmacology, 72, pp.1135-1142.
Bandekar, M., 2012. Development of pharmacovigilance programme for adverse drug reaction monitoring in psychiatry and tuberculosis (Doctoral dissertation, SNDT Women’s University).
Edwards, I.R. and Aronson, J.K., 2015. Adverse drug reactions: definitions, diagnosis, and management. The Lancet, 356(9237), pp.1255-1259.
Alomar, M.J., 2014. Factors affecting the development of adverse drug Reactions. Saudi Pharm J, 22: 83–94.
Chang, Y.P., Huang, S.K., Tao, P. and Chien, C.W., 2012. A population-based study on the association between acute renal failure (ARF) and the duration of polypharmacy. BMC nephrology, 13, pp.1-7.
Farcas A, Bojita M. Adverse drug reactions in clinical practice: a causality assessment of a case of drug-induced pancreatitis. Journal of Gastrointestin Liver Dis. 18(3); 2009: 353-358 Available from: URL: http://www.ncbi.nlm.nih.gov /pubmed/19795031
Shakya, R., Rao, B.S. and Shrestha, B., 2004. Incidence of hepatotoxicity due to antitubercular medicines and assessment of risk factors.
Angamo, M.T., Chalmers, L., Curtain, C.M. and Bereznicki, L.R., 2016. Adverse-drug-reaction-related hospitalisations in developed and developing countries: a review of prevalence and contributing factors. Drug safety, 39, pp.847-857.
Alomar, M.J., 2014. Factors affecting the development of adverse drug reactions. Saudi Pharmaceutical Journal, 22(2), pp.83-94.
Brabete, A.C., Greaves, L., Maximos, M., Huber, E., Li, A. and Lê, M.L., 2022. A sex-and gender-based analysis of adverse drug reactions: A scoping review of pharmacovigilance databases. Pharmaceuticals, 15(3), p.298.
da Silva, K.D.L., Fernandes, F.E.M., de Lima Pessoa, T., Lima, S.I.V.C., Oliveira, A.G. and Martins, R.R., 2019. Prevalence and profile of adverse drug reactions in high-risk pregnancy: a cohort study. BMC Pregnancy and Childbirth, 19(1), pp.1-6.
Kacevska, M., Ivanov, M. and Ingelman‐Sundberg, M.J.C.P., 2011. Perspectives on epigenetics and its relevance to adverse drug reactions. Clinical Pharmacology & Therapeutics, 89(6), pp.902-907.
Volpe, M.R., Bush, R.A.B., Johnson Jr, G.A. and Kwok, C.M., 2008. Barriers to Participation: Challenges faced by members of underrepresented racial and ethnic groups in entering, remaining, and advancing in the ADR field. Fordham Urb. LJ, 35, p.119.
Onder, G., Landi, F., Della Vedova, C., Atkinson, H., Pedone, C., Cesari, M., Bernabei, R., Gambassi, G. and Investigators of the GIFA Study, 2002. Moderate alcohol consumption and adverse drug reactions among older adults. Pharmacoepidemiology and drug safety, 11(5), pp.385-392.
Zopf, Y., Rabe, C., Neubert, A., Hahn, E.G. and Dormann, H., 2008. Risk factors associated with adverse drug reactions following hospital admission: a prospective analysis of 907 patients in two German university hospitals. Drug Safety, 31, pp.789-798.
Rawal, G., Yadav, S., Kumar, R. and Wani, U.R., 2016. Ofloxacin induced angioedema: a rare adverse drug reaction. Journal of Clinical and Diagnostic Research: JCDR, 10(11), p.FD03.
Sahu, R.K., Yadav, R., Prasad, P., Roy, A. and Chandrakar, S., 2014. Adverse drug reactions monitoring: prospects and impending challenges for pharmacovigilance. Springerplus, 3(1), pp.1-9.
Kumar, M.M., Assessment of Adverse Drug Reactions and Evaluation of Possible Predisposing Factors.
Srikanth, M.S. and Adepu, R., 2014. Adverse drug reaction monitoring and reporting by community pharmacists: a review. World Journal of Pharmaceutical Research, 3(8), pp.1223-33.
Basak, S.C. and Sathyanarayana, D., 2010. Evaluating medicines dispensing patterns at private community pharmacies in Tamilnadu, India. Southern Med Review, 3(2), pp.27-31.
Tobaiqy, M., Stewart, D., Helms, P.J., Bond, C.M., Lee, A.J. and McLay, J., 2010. Views of parents and pharmacists following participation in a paediatric pharmacovigilance study. Pharmacy world & science, 32, pp.334-338.
Srikanth, M.S. and Adepu, R., 2019. Assessment of structured educational intervention and barriers of community pharmacists towards adverse drug reaction reporting and monitoring in South Indian district. Drug Invention Today, 12.
Rajalakshmi, R., Devi, B.V., Prasad, T.D., Swetha, S. and Dharini, B., 2017. Knowledge, attitude and practice towards pharmacovigilance and adverse drug reaction reporting among nurses in a tertiary care hospital, Tirupati. International Journal of Pharmaceutical and Clinical Research, 9(11), pp.683-689.
Coleman, J.J. and Pontefract, S.K., 2016. Adverse drug reactions. Clinical Medicine, 16(5), p.481.
Reumerman, M., Tichelaar, J., Piersma, B., Richir, M.C. and Van Agtmael, M.A., 2018. Urgent need to modernize pharmacovigilance education in healthcare curricula: review of the literature. European Journal of Clinical Pharmacology, 74, pp.1235-1248.
Schnipper, J.L., Kirwin, J.L., Cotugno, M.C., Wahlstrom, S.A., Brown, B.A., Tarvin, E., Kachalia, A., Horng, M., Roy, C.L., McKean, S.C. and Bates, D.W., 2006. Role of pharmacist counseling in preventing adverse drug events after hospitalization. Archives of internal medicine, 166(5), pp.565-571.
Hadi, M.A., Neoh, C.F., Zin, R.M., Elrggal, M.E. and Cheema, E., 2017. Pharmacovigilance: pharmacists’ perspective on spontaneous adverse drug reaction reporting. Integrated Pharmacy Research and Practice, pp.91-98.
Cite This Work
To export a reference to this article please select a referencing stye below:
Academic Master Education Team is a group of academic editors and subject specialists responsible for producing structured, research-backed essays across multiple disciplines. Each article is developed following Academic Master’s Editorial Policy and supported by credible academic references. The team ensures clarity, citation accuracy, and adherence to ethical academic writing standards
Content reviewed under Academic Master Editorial Policy.
- Editorial Staff
- Editorial Staff




